Pfizer Inc. is recalling 1 million packets of birth control pills because of a packaging error that could leave women with an inadequate dose of the hormone-based drugs and raise the risk that they will get pregnant accidentally.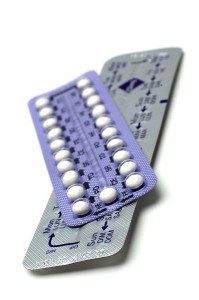
The news came a day after the drug giant and New York-based maker of Viagra said its fourth-quarter profit fell by half and lowered its 2012 forecast due to other business issues.
Pfizer found that some packets of the drugs had too many active tablets, while others had too few. Oral birth control products use a series of 21 drug tablets and 7 inactive sugar tablets to regulate the menstrual period while providing contraception.
A company spokeswoman said the problem was caused by both mechanical and visual inspection failures on the packaging line. She said the problem has been corrected.
The recall is not related to safety but does raise the risk of unintended pregnancy.
The drugs were distributed to warehouses, clinics and retail pharmacies throughout the U.S.
The recall affects 14 lots of Lo/Ovral-28 tablets and 14 lots of generic Norgestrel and Ethinyl Estradiol tablets, which are marketed in the U.S. under the Akrimax Pharmaceuticals brand.
Was this article valuable?
Here are more articles you may enjoy.

 Iran Starts Bitcoin-Backed Ship Insurance for Hormuz Strait
Iran Starts Bitcoin-Backed Ship Insurance for Hormuz Strait  Lake Tahoe Power Crunch Shows AI’s Growing Energy Toll in West
Lake Tahoe Power Crunch Shows AI’s Growing Energy Toll in West  34,000 PG&E Customers Without Power on California Fire Risk
34,000 PG&E Customers Without Power on California Fire Risk  Snap, YouTube Settle School-Social Media Suit Ahead of Trial
Snap, YouTube Settle School-Social Media Suit Ahead of Trial 